
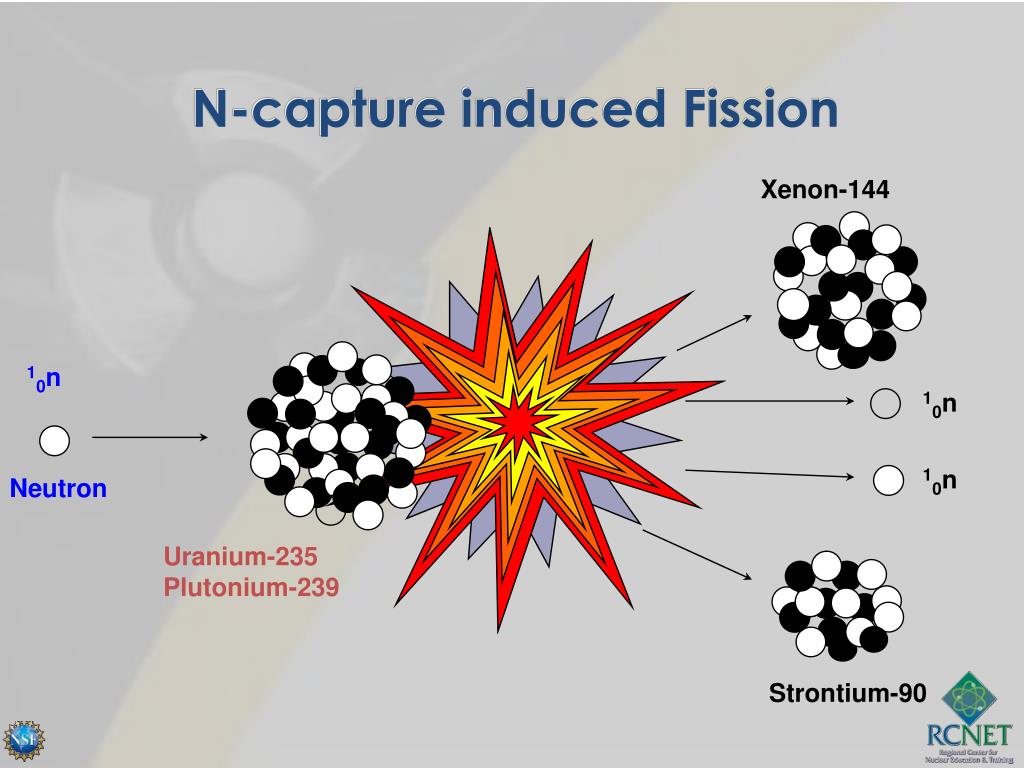
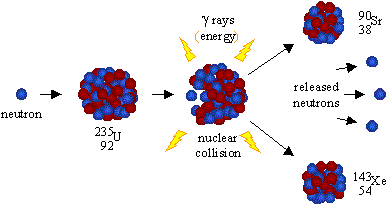
In an old system (≫500 ky) a radioactive secular equilibrium is established between the parent 238U or 235U and their daughter radioisotopes. Uranium-238 and Uranium-235 are the parent isotopes of decay chains that can be used to provide a chronology back to ∼500 ky.
Pike, in Encyclopedia of Analytical Science (Second Edition), 2005 Uranium-Series Dating The combined capacity of these plants was about 40 MSWU/y at the end of 2000. Gaseous diffusion plants are known to exist in Argentina, China, France, Russia and the United States. Improved technology may reduce this somewhat. An annual production of 10 MSWU requires an installed capacity of ∼ 2900 MW in present plants, or ∼ 2500 kWh SWU −1. Isotope separation through gaseous diffusion is a very energy-consuming process due to the compression work and cooling required. In more recent designs the concentration of 235U in the waste is increased to ∼ 0.3% to minimize cost. With a waste flow in which the isotopic abundance of 235U is 0.2%, (2.48) shows that for each mole of product obtained 156 moles of feed are necessary. In §2.8.1 the number of stages and the interstage flow relative to the product flow was given for enrichment of 235U from its natural isotopic abundance of 0.71% to a value of 80%. A 1 GWe nuclear light water reactor station requires about 180 × 10 3 SWU in initial fueling and then 70 – 90 × 10 3 SWU for an annual reload. The cost of isotope separation is obtained by assigning a value to one separative work mass unit (kgSW or SWU). (2.57) V ( x i ) = ( 2 x i − 1 ) ln Īs seen from (2.56), separative work has the dimension of mass, and can be thought of as the mass flow rate multiplied by the time required to yield a given quantity of product. Water decomposes UF 6 according to the equation The membrane must be inert to attack by UF 6.
UF 6 is highly corrosive and attacks most materials. UF 6 sublimes at 64☌, which means that the separation process must be conducted at a temperature above this. Moreover, the membranes must have good mechanical stability to withstand the pressure difference across them. In order that large gas volumes can flow through the membrane, millions of pores are required for each square centimeter. 10 – 100 nm in diameter) in order to obtain isotopic separation. The cells are divided into two parts by a membrane which must have very small pores (e.g. The following conditions must be considered in the technical application of the separation. For the isotopic molecules 235UF 6 and 238UF 6, a value of 1.0043 is theoretically possible for α (cf. In addition to its volatility, UF 6 has the advantage that fluorine consists of only one isotope, 19F. 235U is enriched through gaseous diffusion using the relatively volatile uranium compound UF 6.
